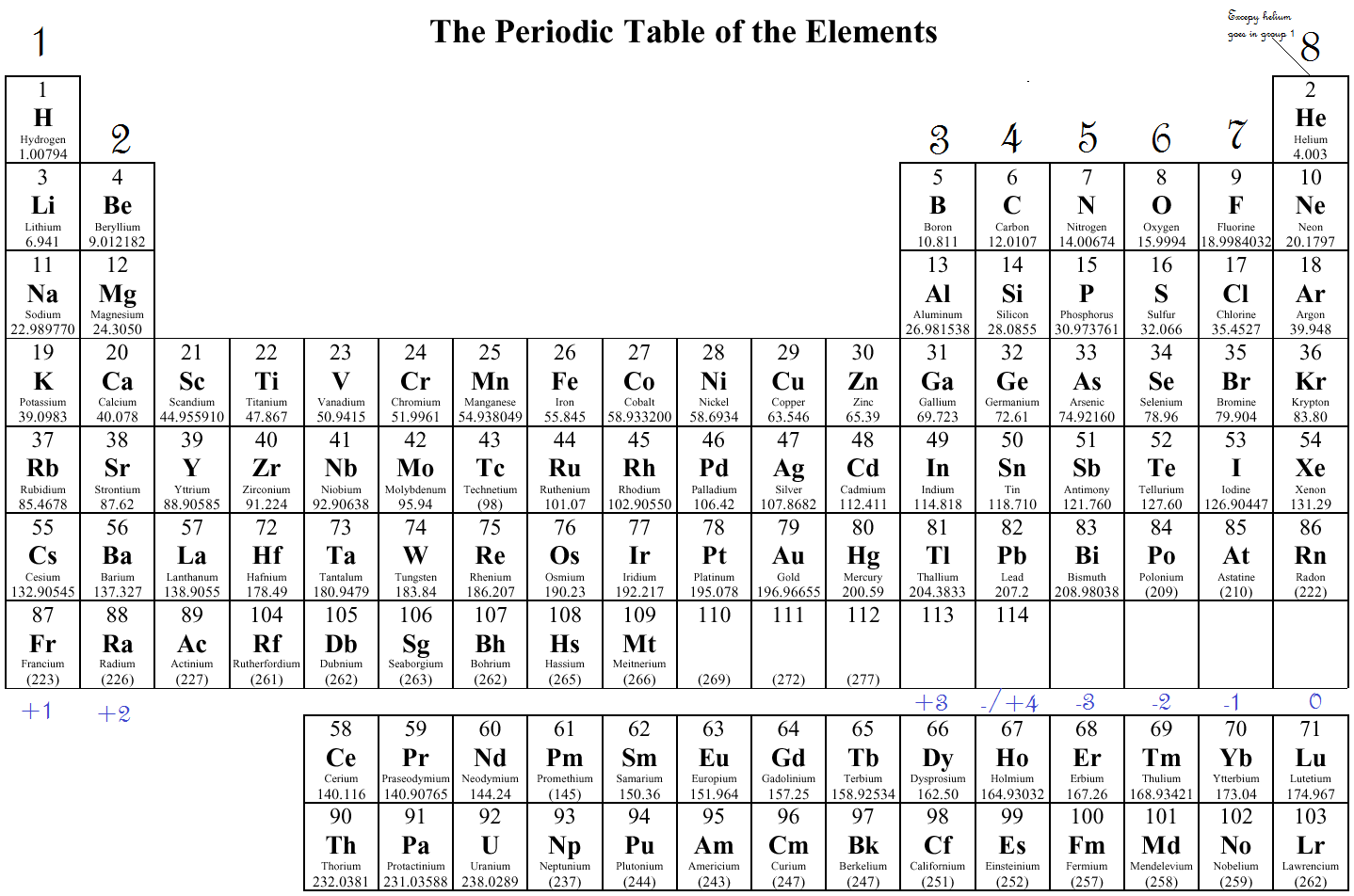
Using Cannizzaro's atomic weights, Mendeleev and Lothar Meyer made a great discovery, the periodic law: If you arrange the elements by their atomic weights, there is a periodic repetition in properties such as valence. aluminum oxide, Al 2O 3: aluminum has a valence of 3īy the 1860s, ~60 elements were known.hydrogen sulfide, H 2S: sulfur has a valence of 2.lithium oxide, Li 2O: lithium has a valence of 1.methane, CH 4: carbon atoms have a valence of 4.Valence is the number of connections an atom tends to form. Scientists soon observed patterns in the valence of the different elements. Once Cannizzaro convinced most chemists to accept it, chemists were able to study the actual atomic weights and formulas. Avogadro's hypothesis let chemists figure out atomic weights and formulas together.
His paper explaining how to calculate molecular weights was distributed to everyone, including Julius Lothar Meyer, who wrote that when he read it "doubts disappeared and a feeling of quiet certainty took their place". Eventually, Cannizzaro revived Avogadro's hypothesis at a big meeting of chemists in 1860. However, Berzelius, who was working hardest on this problem, didn't believe either Avogadro or Faraday.īerzelius' beliefs held up the progress of science for about 50 years. The other was Faraday's law, which measured masses of elements produced by a set amount of current. One was Avogadro's hypothesis based on Gay-Lussac's law, which allowed chemists to relate the equivalent masses to equivalent volumes. There were two good ways available to figure this out. But what about elements? For instance, what mass of element A reacts with 1 g of element B? Knowing these numbers could be very useful, but it was hard to relate the equivalent masses to the actual masses of the atoms because they didn't know the formulas. For instance, what mass of acid A neutralizes base B? These numbers were easy to measure and practically useful. Many chemists were interested in knowing the "chemical equivalents" of different substances. Understand and describe how the periodic table was first organized.
